Semax is a synthetic heptapeptide derived from an adrenocorticotropic hormone fragment and researched for its effects on neuronal network activity, neurotrophins (e.g., BDNF), inflammation signaling, and oxidative stress pathways. While Semax has clinical history in Russia, the English-language human evidence base remains limited, so it should be approached with clear expectations, careful screening, and clinician-guided risk–benefit review. Learn more about Peptide Therapy.
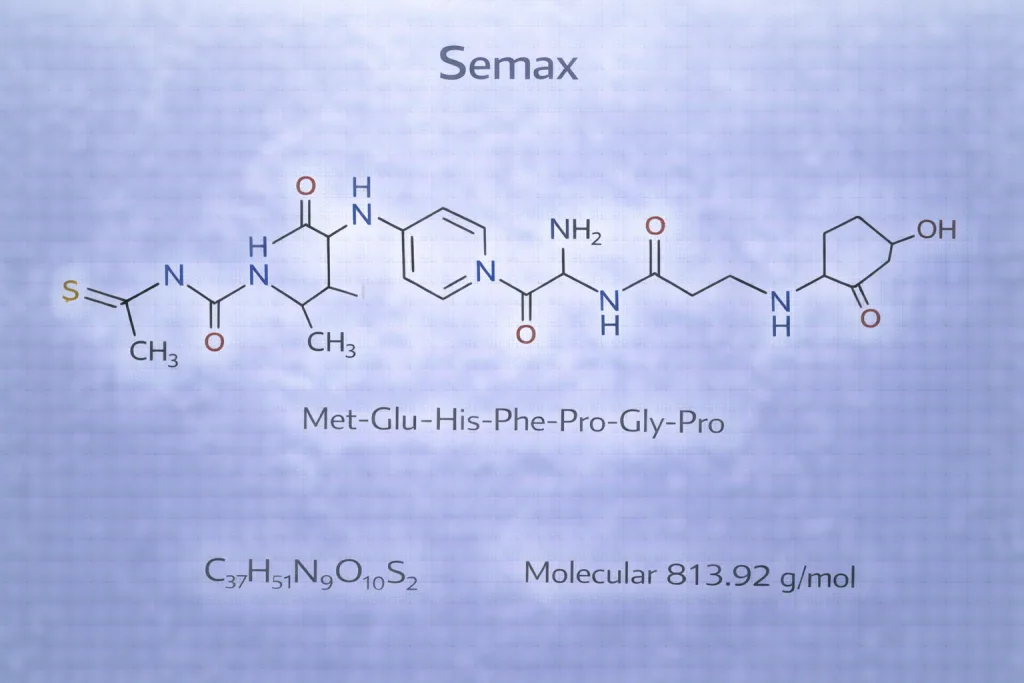
Class: ACTH fragment analog (“melanocortin” derivative)
Often referenced as ACTH(4–7)PGP / ACTH(4–10) analog in the literature
Sequence: Met–Glu–His–Phe–Pro–Gly–Pro
Molecular formula / MW: C₃₇H₅₁N₉O₁₀S, ~813.9 g/mol
“More stable” variant commonly discussed: N-acetyl Semax (N-terminal acetylation; marketed as a stability-modified form—high-quality human comparative data is limited).
Studied / plausible use-cases (stronger preclinical support): neuroprotection in ischemia models, neurotrophin modulation (e.g., BDNF), immune/inflammation gene expression changes in stroke models.
Limited human data: small intranasal study showing measurable changes in resting-state network findings shortly after dosing.
Common “nootropic” positioning (lower certainty): focus, concentration, stress support, mood resilience—often discussed clinically, but not backed by large modern RCTs in major English-language literature.
Regulatory note (US): Semax is not FDA-approved; quality, dosing, and sterility depend on sourcing/compounding practices.
Avoid / caution: pregnancy/lactation; acute psychosis; significant anxiety disorders; seizure history (listed as contraindications in translated product leaflet materials).
Safety reality check: published human safety data is not as robust as for FDA-approved therapies; adverse effects may be underreported outside regulated trials.
Start with a Functional Medicine Evaluation — review sleep, labs, metabolic and inflammatory drivers, and build a cognition-focused plan.
Individualized care • Medically supervised • Lab-guided treatment
Intranasal solutions/drops are commonly referenced in international markets; translated labeling describes 0.1% and 1% nasal formulations and provides per-drop dosing details.
Lyophilized powder formats exist in research/peptide channels; there is no FDA-standardized US label to anchor concentration, sterility standards, or administration protocols.
A small placebo-controlled study evaluated intranasal 1% Semax with imaging performed minutes after dosing.
Translated leaflet guidance for 0.1% nasal drops notes 50 mcg per drop and describes intranasal administration with a dropper (titration based on clinical context).
SubQ protocols are commonly discussed in peptide practice settings, but high-quality published dosing standards are limited in widely indexed English-language clinical trials. If used, rely on pharmacy labeling + clinician protocol, and monitor tolerability closely. (Evidence-limited compared with intranasal.)
Because Semax research frequently intersects with neuroinflammation/immune signaling, hs-CRP can be a practical, inexpensive marker to trend alongside sleep, stress load, cognition, and recovery metrics—especially in a functional medicine optimization plan.
When a clinician is evaluating cognitive performance concerns, stress resilience, or post-neurologic recovery goals after foundational drivers are addressed (sleep, metabolic health, micronutrients, thyroid, mood, medications).
When the plan includes objective tracking (symptom scales, sleep metrics, cognitive tasks, BP/HR, side effects) and a clear stop-rule if benefits aren’t measurable.
Evidence gradient matters: Strong mechanistic/preclinical signal ≠ proven clinical outcomes in broad populations.
Mechanisms are multi-pathway: Neurotrophin effects (BDNF), immune gene expression shifts, and receptor-level melanocortin activity have all been described; clinical significance varies by context.
Drug–condition matching: Screen carefully for anxiety spectrum disorders, seizure risk, pregnancy/lactation, and psychiatric instability.
Semax is a synthetic ACTH-fragment heptapeptide researched for neurotrophic, neuroprotective, and immunomodulatory signaling. Mechanistic studies show associations with BDNF changes and altered inflammatory/neurotransmission gene expression in ischemia models, and a small human study suggests acute effects on brain network measures after intranasal dosing. Clinically, it has historical use in Russia, but for US audiences the key limitation is the lack of large, high-quality randomized human trials for most “nootropic” claims—so responsible use centers on individualized screening, conservative dosing strategy, and outcomes tracking.
Learn about alternative peptide therapies such as N Acetyl Selank Peptide for mood regulation or cognitive benefits. Semorelin Peptide is an excellent treatment option for muscle growth and repair, Pentadeca Arginate for recovery, Pinealon peptide for its cognitive benefits, and/or NAD+ for longevity.
Preclinical Studies – Cerebral Ischemia:
PMID: 34201112 – Brain Protein Expression Profile Confirms the Protective Effect of Semax in Cerebral Ischemia-Reperfusion (2021)
PMID: 32580520 – Novel Insights Into the Protective Properties of Semax at the Transcriptome Level (2020)
PMID: 28255762 – Semax Regulates Expression of Immune Response Genes During Ischemic Brain Injury (2017)
PMID: 24661604 – Semax Affects Expression of Genes Related to Immune and Vascular Systems in Focal Ischemia (2014)
BDNF and Neurotrophin Studies:
PMID: 16635254 – Semax Binds Specifically and Increases BDNF Protein in Rat Basal Forebrain (2006)
PMID: 19662538 – Comparison of NGF and BDNF Gene Expression Under Semax Action (2010)
PMID: 17353092 – Neurotrophin Gene Expression in Rat Brain Under Semax Action (2007)
Other Mechanisms:
PMID: 40692165 – Semax Targets μ Opioid Receptor for Functional Recovery After Spinal Cord Injury (2025)
PMID: 25310602 – Semax Has High Affinity for Copper(II) and Protective Ability Against Metal Toxicity (2015)
PMID: 35080861 – Semax Affects Copper-Induced Aβ Aggregation in Alzheimer’s Models (2022)The Peptide Semax Affects the Expression of Genes Related to the Immune and Vascular Systems in Rat Brain Focal Ischemia: Genome-Wide Transcriptional Analysis.
Schedule a Peptide Therapy Consult — discuss evidence level, safety screening, dosing strategy, and measurable outcomes tracking.
Personalized care • Clinician-directed • Data-driven treatment
Business Hours
Monday: 9AM – 6PM
Tuesday: 9AM – 6PM
Wednesday: 9AM – 6PM
Thursday: 9AM – 6PM
Friday: 9AM – 6PM
Saturday: Closed
Sunday: Closed

Comprehensive care with a whole person approach, specializing in injury management, wellness, and longevity.
3874 San Jose Park Drive
Suite 5
Jacksonville, Florida 32217
Disclaimer: The content on this website is for informational purposes only and does not constitute medical advice or replace professional medical care, diagnosis, or treatment. Eligibility for medical treatments will be determined by your healthcare provider, who will exercise discretion regarding your treatment plan. All images are intended for educational purposes only. Statements made on this website have not been evaluated by the Food and Drug Administration. Always consult your healthcare provider before initiating or discontinuing any treatment.
Intercoastal Health – Copyright © 2026 All Rights Reserved - Privacy Policy